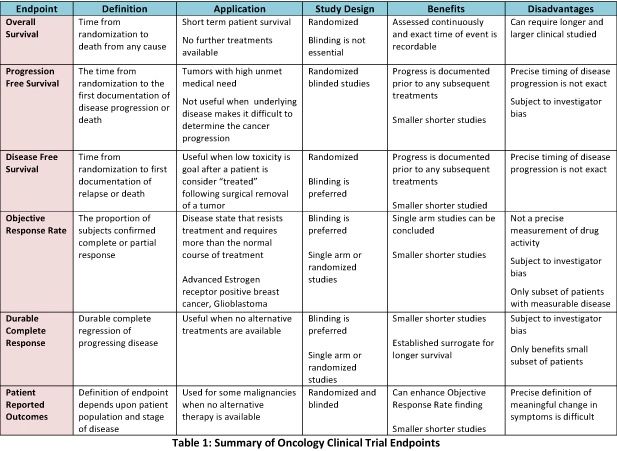
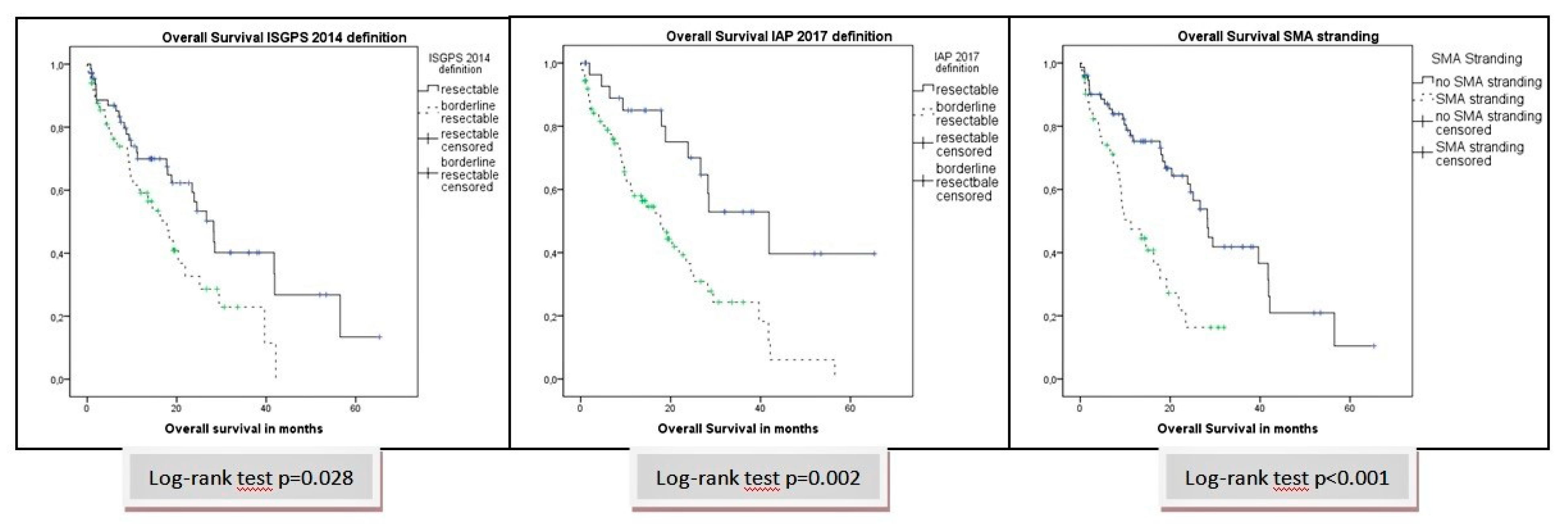
A novel high-risk stage II colon cancer classification method may improve prognosis with patients who receive adjuvant chemotherapy

Progression-Free Survival: What Does It Mean for Psychological Well-Being or Quality of Life? - NCBI Bookshelf
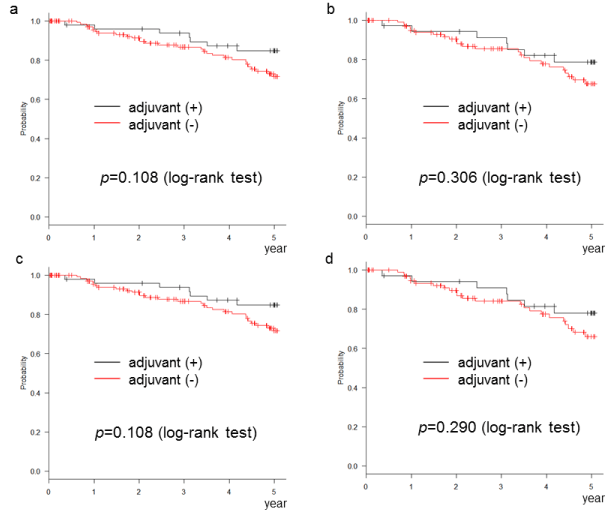
Cancers | Free Full-Text | Systematic Analysis of Accuracy in Predicting Complete Oncological Resection in Pancreatic Cancer Patients—Proposal of a New Simplified Borderline Resectability Definition | HTML

The Association Between Radiation Therapy Dose and Overall Survival in Patients With Intracranial Infiltrative Low-Grade Glioma Treated With Concurrent and/or Adjuvant Chemotherapy - Advances in Radiation Oncology

References in Outcomes and endpoints in trials of cancer treatment: the past, present, and future - The Lancet Oncology

Definition of progression free survival and overall survival for the... | Download Scientific Diagram

Definition of long (> 12 mo) and short (≤ 12 mo) survival patterns from... | Download Scientific Diagram

Heterogeneity in Definitions of High-risk Prostate Cancer and Varying Impact on Mortality Rates after Radical Prostatectomy - European Urology Oncology

Prostate-specific membrane antigen in circulating tumor cells is a new poor prognostic marker for castration-resistant prostate cancer

Event-free and overall survival following neoadjuvant weekly paclitaxel and dose-dense AC +/- carboplatin and/or bevacizumab in triple-negative breast. - ppt download

The new ESPEN diagnostic criteria for malnutrition predict overall survival in hospitalised patients - Clinical Nutrition

Fifteen-Year Biochemical Relapse-Free Survival, Cause-Specific Survival, and Overall Survival Following I125 Prostate Brachytherapy in Clinically Localized Prostate Cancer: Seattle Experience - International Journal of Radiation Oncology, Biology, Physics

Guidelines for time-to-event end point definitions in sarcomas and gastrointestinal stromal tumors (GIST) trials: results of the DATECAN initiative (Definition for the Assessment of Time-to-event Endpoints in CANcer trials) – topic of
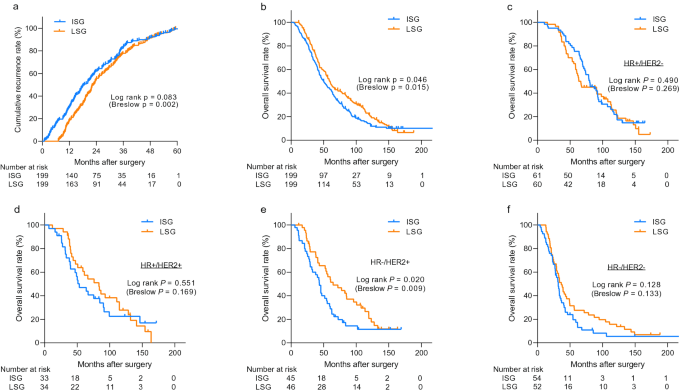
![PDF] Incorporating Clinical Trial Data Into Daily Cancer Care | Semantic Scholar PDF] Incorporating Clinical Trial Data Into Daily Cancer Care | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/30f4b9adffa198bf79a177643e74ec64b4107203/3-Table1-1.png)