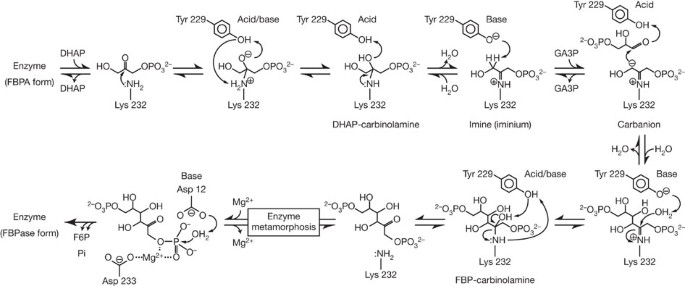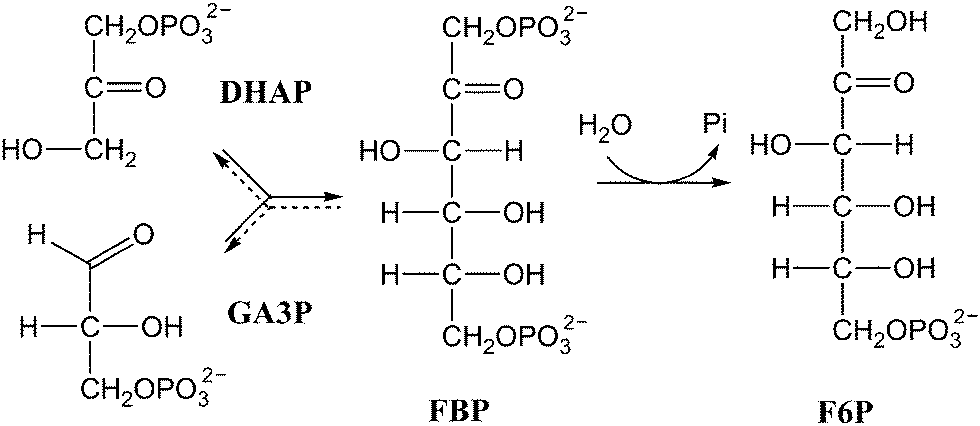
QM/MM studies of the mechanism of unusual bifunctional fructose-1,6-bisphosphate aldolase/phosphatase - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C3CP55263B
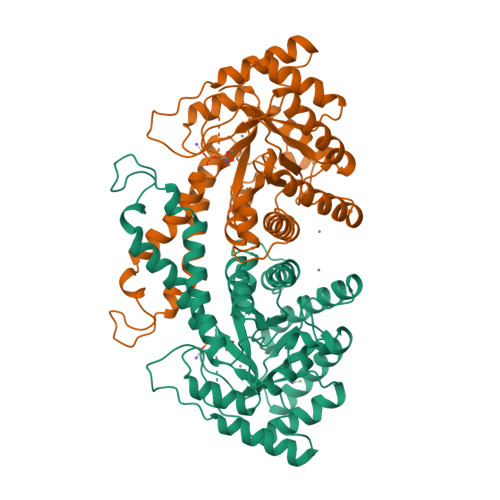
RCSB PDB - 1B57: CLASS II FRUCTOSE-1,6-BISPHOSPHATE ALDOLASE IN COMPLEX WITH PHOSPHOGLYCOLOHYDROXAMATE

Evaluation of four microbial Class II fructose 1,6-bisphosphate aldolase enzymes for use as biocatalysts. | Semantic Scholar

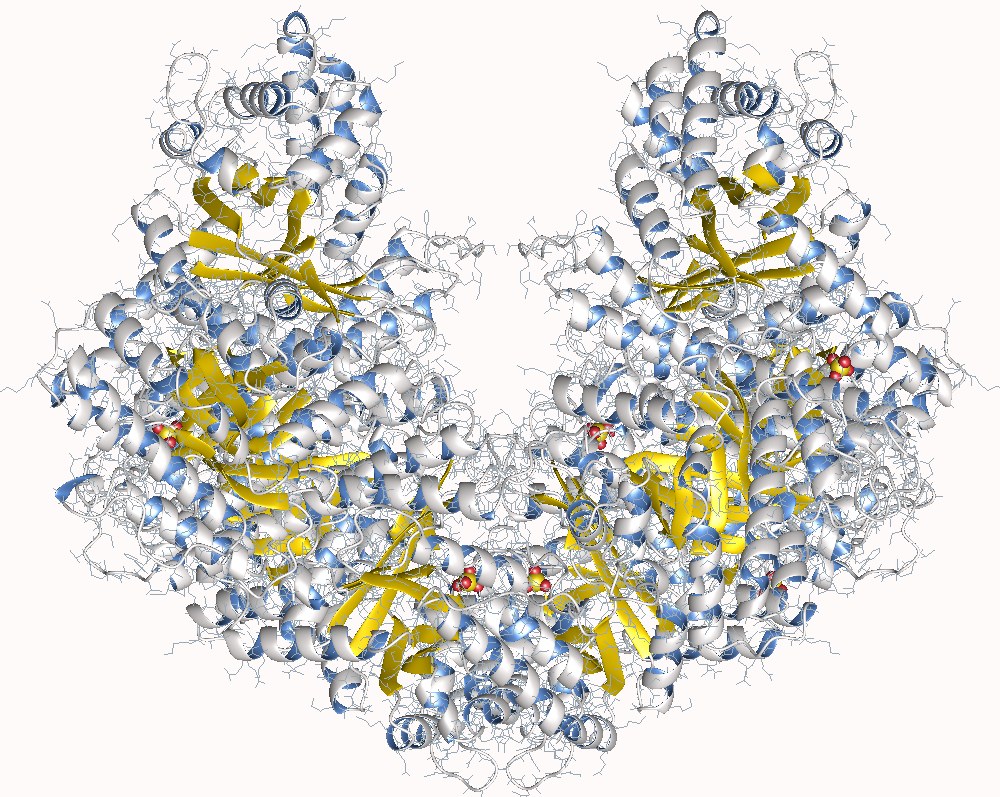
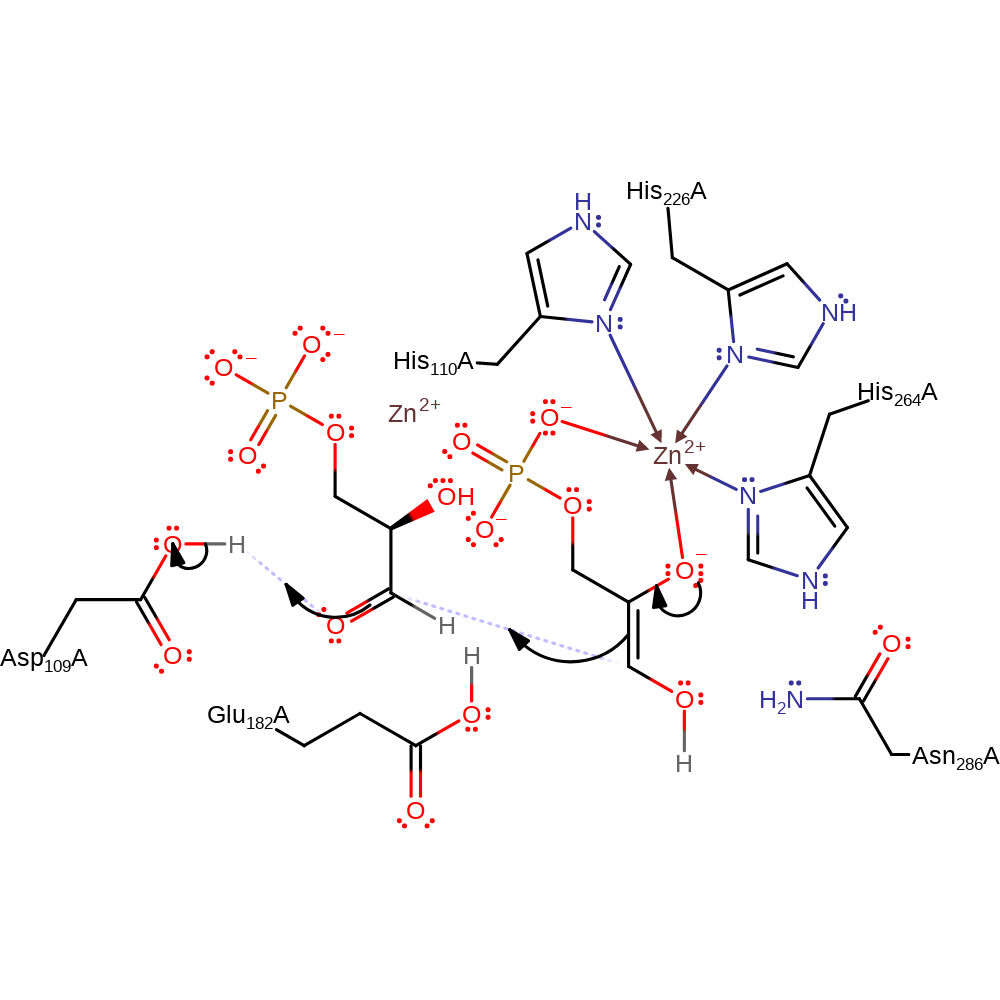
The crystal structure of a class II fructose-1,6-bisphosphate aldolase shows a novel binuclear metal-binding active site embedded in a familiar fold: Structure

Exploring substrate binding and discrimination in fructose1,6‐bisphosphate and tagatose 1,6‐bisphosphate aldolases - Zgiby - 2000 - European Journal of Biochemistry - Wiley Online Library

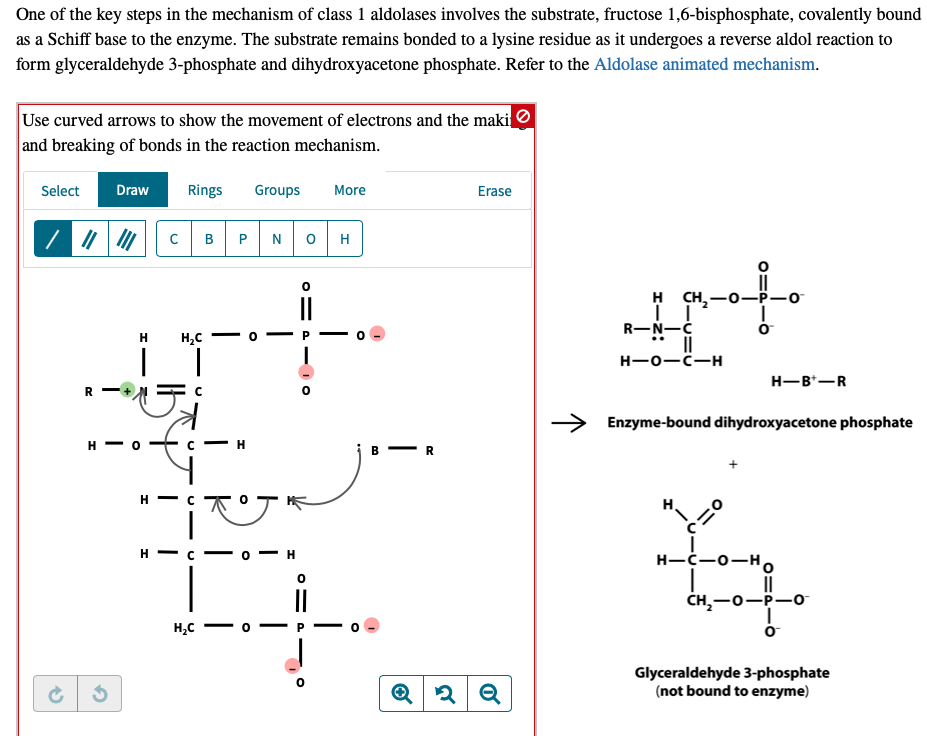
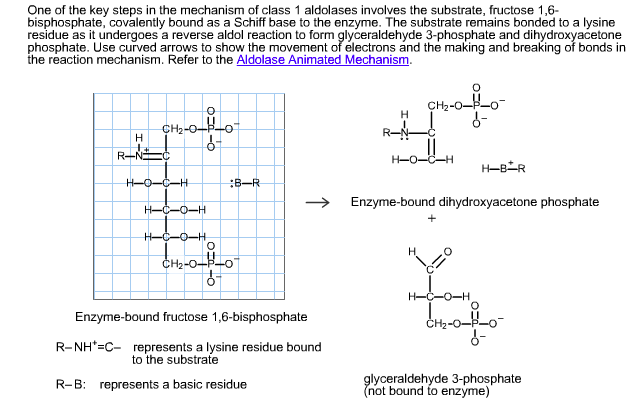
Carbon Isotope Effects on the Fructose-1,6-bisphosphate Aldolase Reaction, Origin for Non-statistical 13C Distributions in Carbohydrates* - Journal of Biological Chemistry