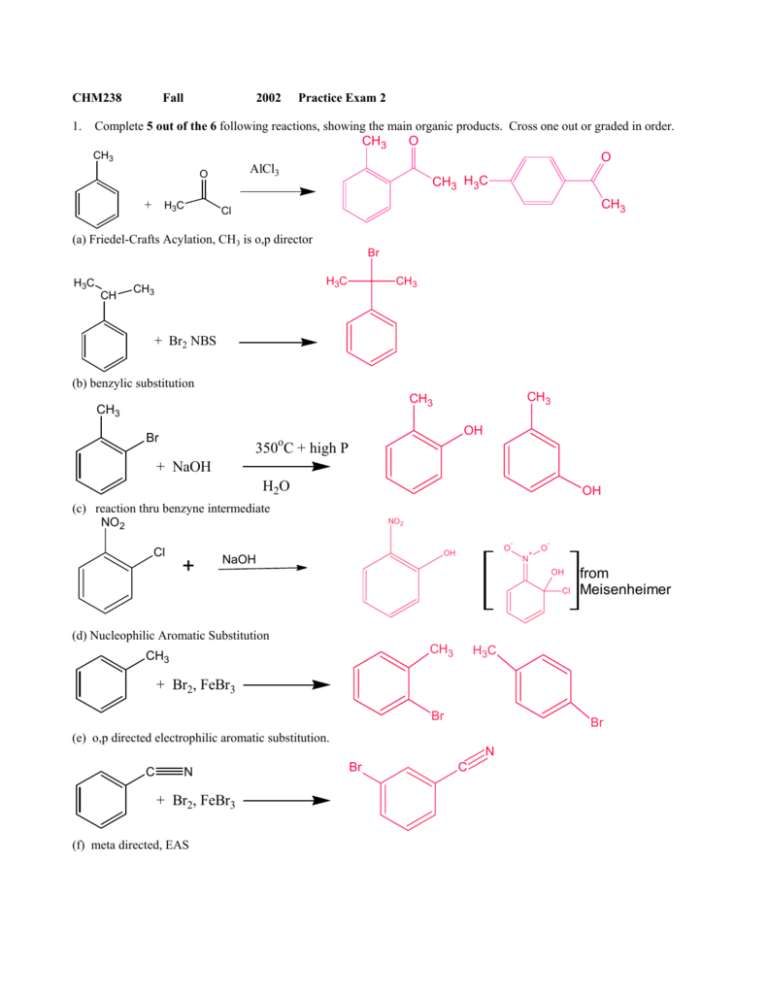
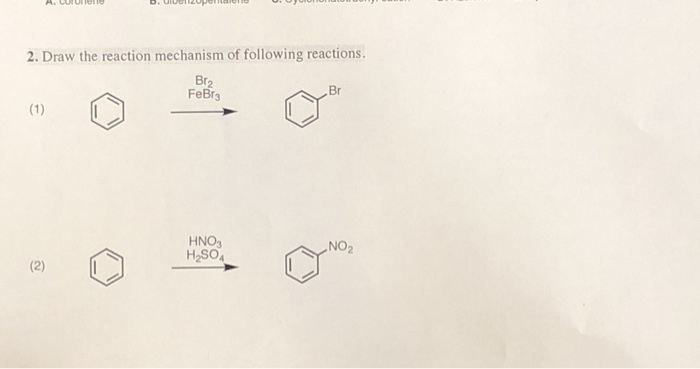
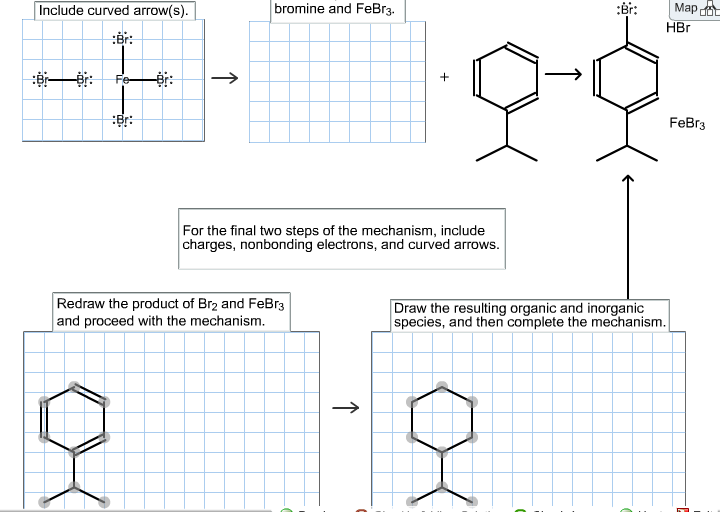
The electrophilic aromatic substitution of isopropylbenzene with FeBr3, Br2 gives 1-bromo-4-isopropylbenzene. Complete the curved-arrow mechanism below,... - HomeworkLib
![Ortho - para - meta directing] In this question, Br2 with FeBr3 was reacted with this ketone. I have a few questions about the answer below : 1-) Why is it o-p Ortho - para - meta directing] In this question, Br2 with FeBr3 was reacted with this ketone. I have a few questions about the answer below : 1-) Why is it o-p](https://preview.redd.it/2qnb7sou3r051.jpg?auto=webp&s=6c003c8bcf00ef4fe3390598987efa770b89f3f0)
Ortho - para - meta directing] In this question, Br2 with FeBr3 was reacted with this ketone. I have a few questions about the answer below : 1-) Why is it o-p
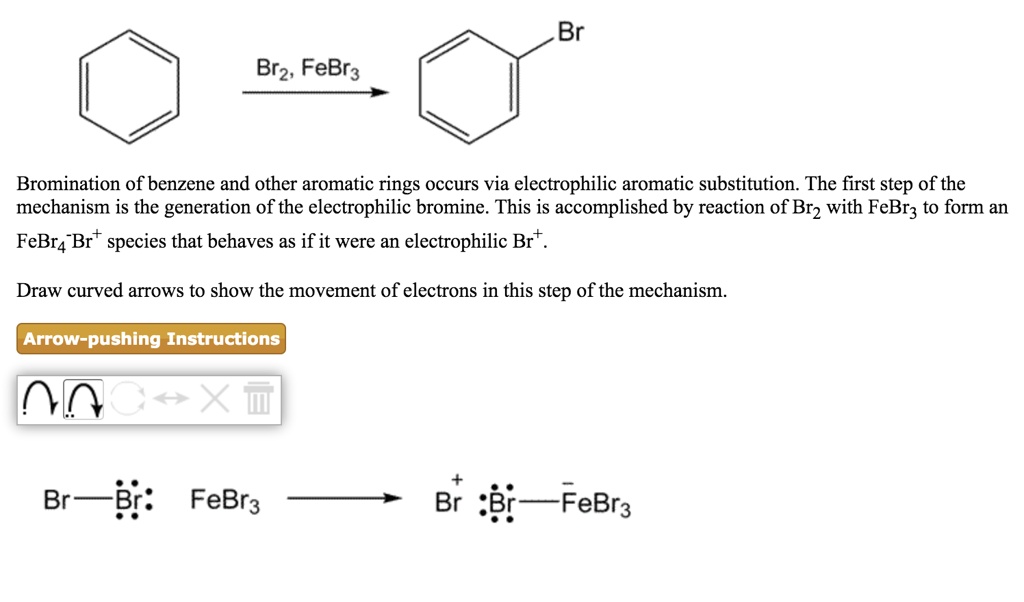
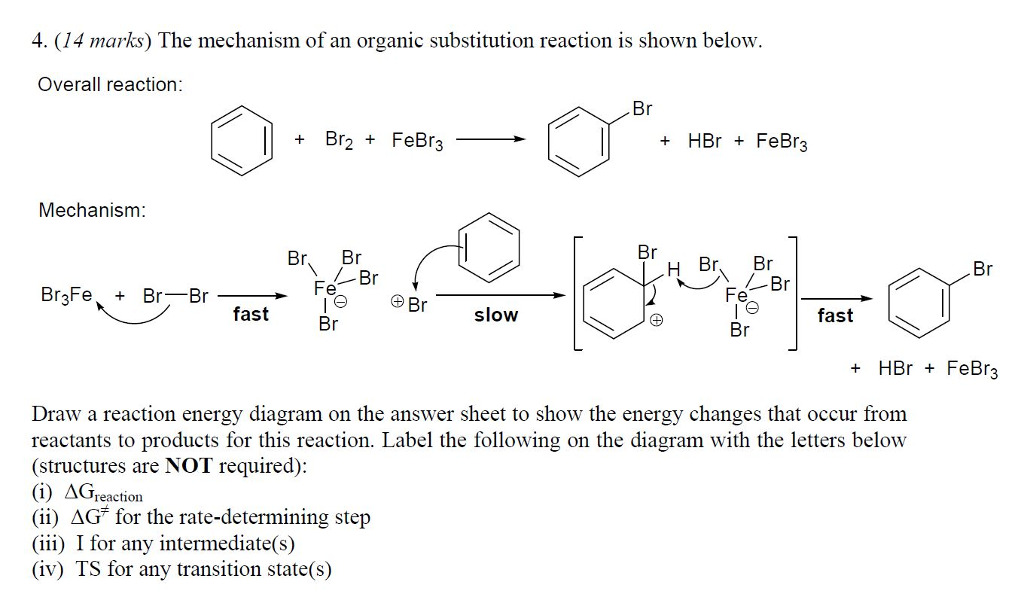
SOLVED:Br Brz' FeBr3 Bromination of benzene and other aromatic rings occurs via electrophilic aromatic substitution. The first step of the mechanism is the generation of the electrophilic bromine This is accomplished by
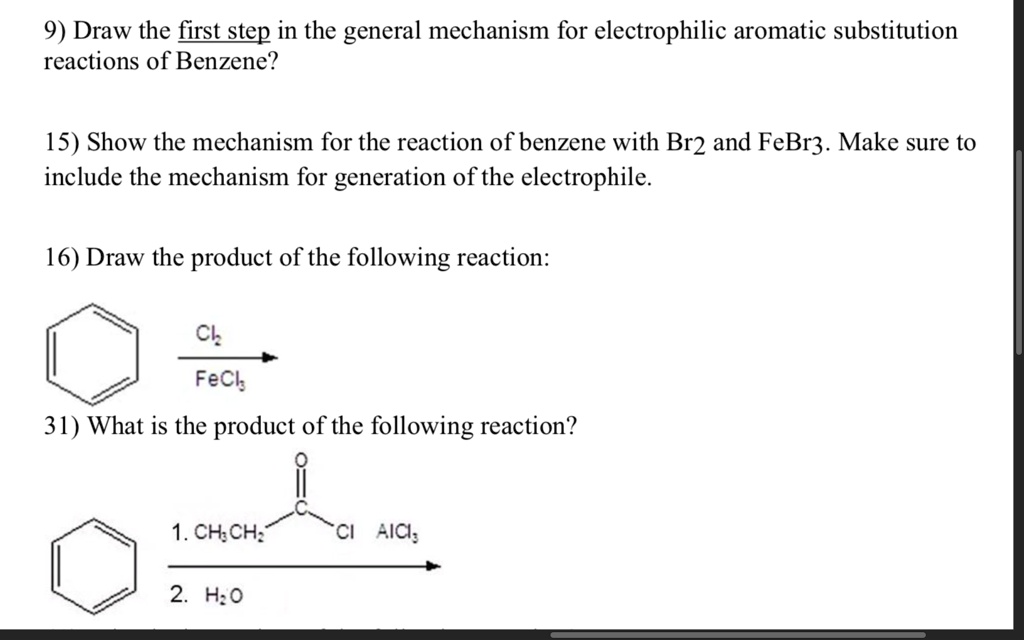
SOLVED:9) Draw the first step in the general mechanism for electrophilic aromatic substitution reactions of Benzene? 15) Show the mechanism for the reaction of benzene with Br2 and FeBr3. Make sure to


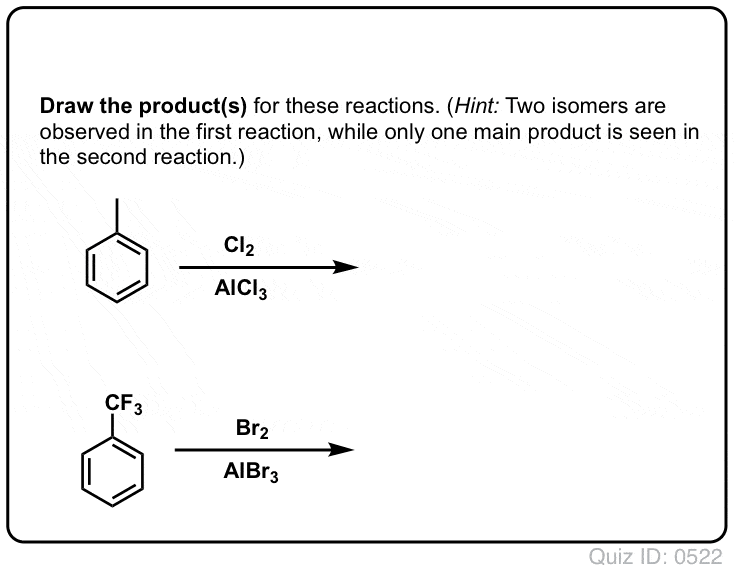
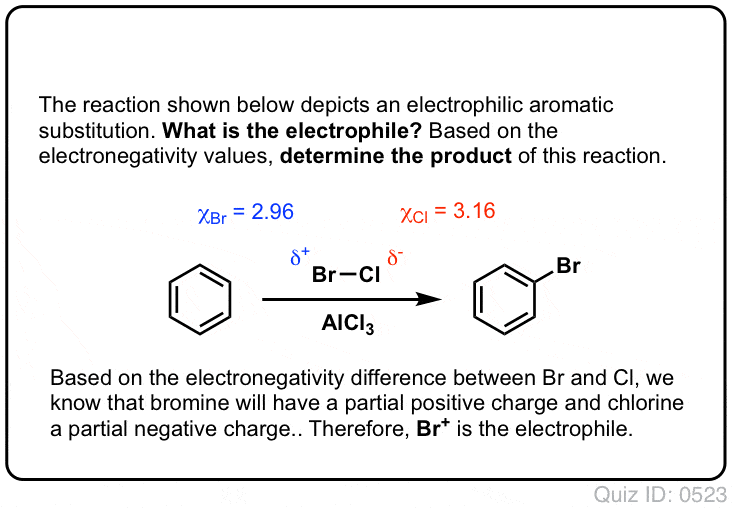
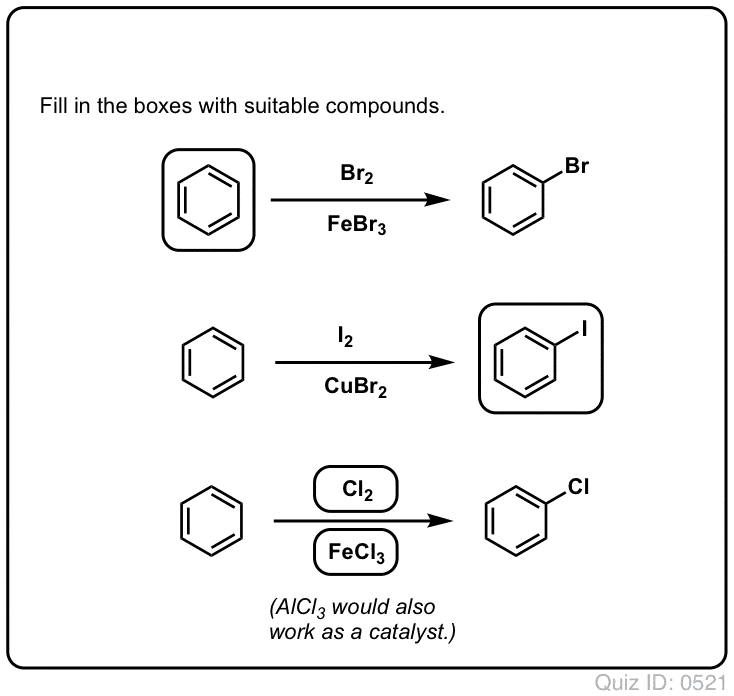
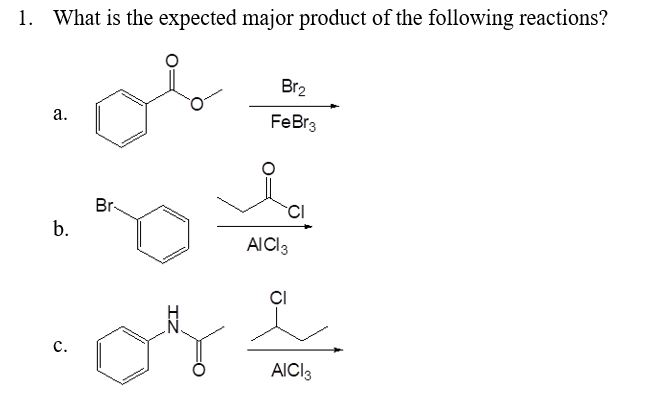



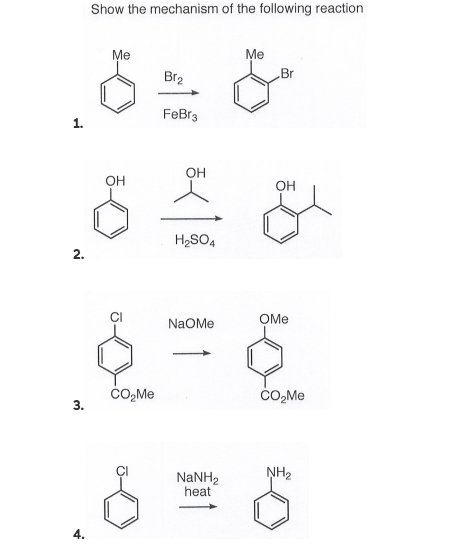

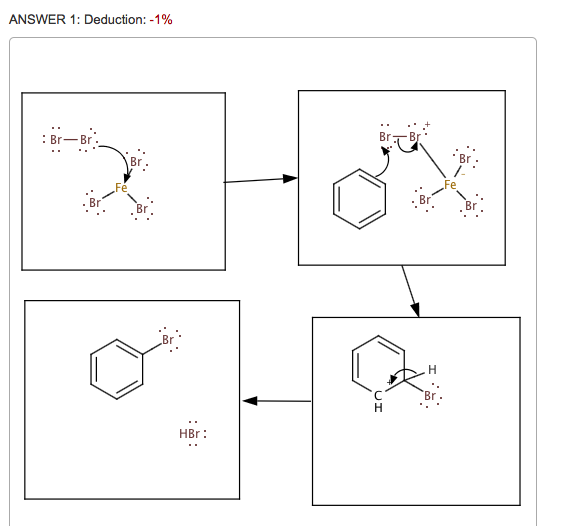


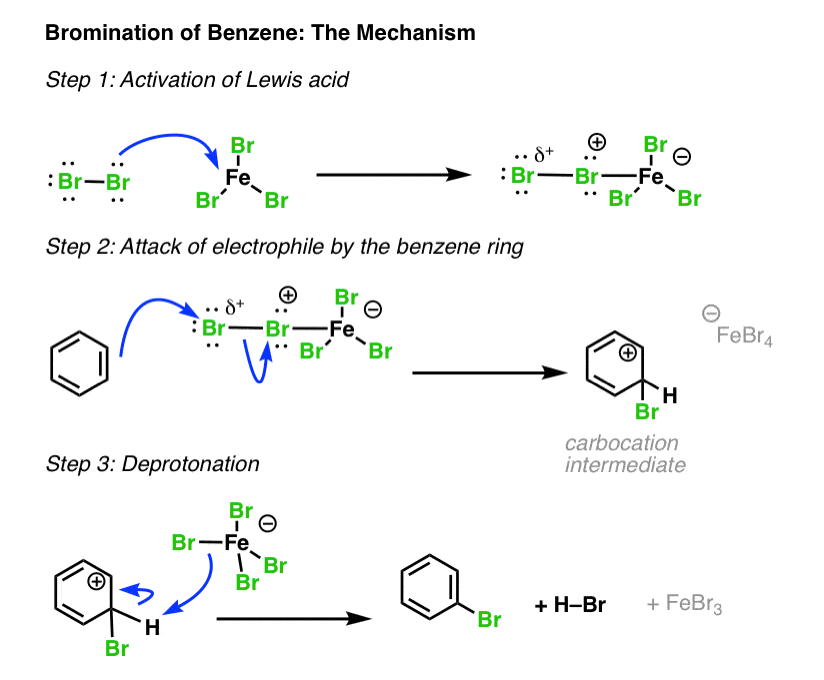
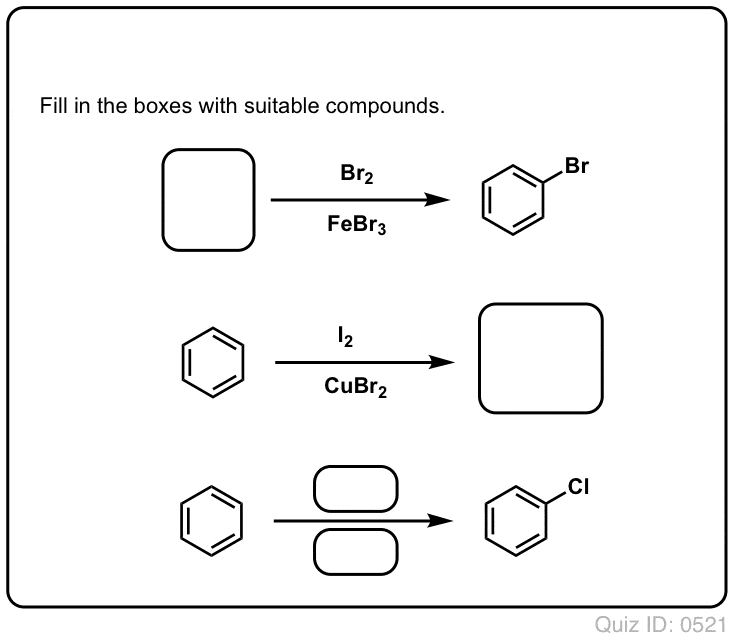


![Bromination [Br2 plus catalyst] - ChemistryScore Bromination [Br2 plus catalyst] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Bromination1-768x379.png)